Australia Regenerative Medicine Market Size, Share, Report 2025-2033
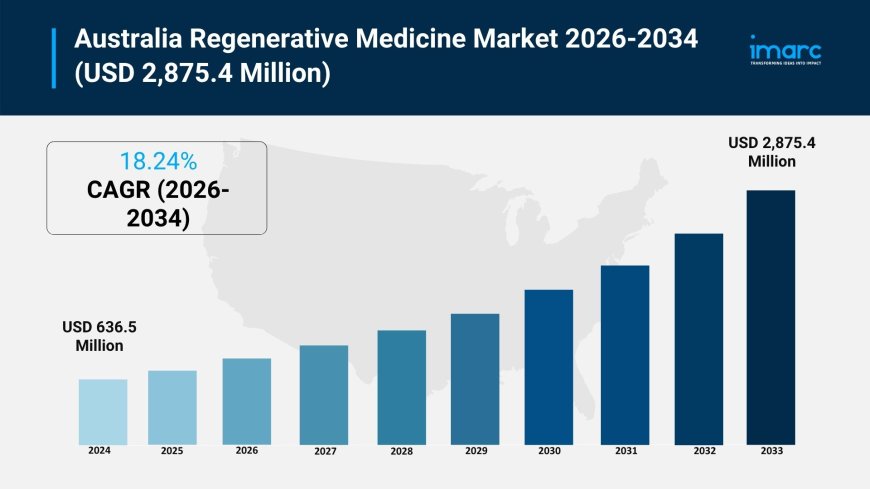
The Australia regenerative medicine market size reached USD 636.5 Million in 2025. Looking forward, IMARC Group expects the market to reach USD 2,875.4 Million by 2034, exhibiting a growth rate (CAGR) of 18.24% during 2026-2034.
Market Overview
The Australia regenerative medicine market size reached USD 636.5 Million in 2025 and is anticipated to grow substantially, reaching USD 2,875.4 Million by 2034. This growth is driven by the rising prevalence of chronic and age-related diseases, advancements in stem cell research, and strong government support for innovation. The focus on personalized medicine, breakthroughs in tissue engineering, and increased R&D investments further propel the market. Enhanced regulatory frameworks and collaboration between academia and industry also contribute to expanding the market share.
How AI is Reshaping the Future of Australia Regenerative Medicine Market:
- AI enables precision medicine by enhancing patient-specific stem cell therapy, reducing immune rejection and improving therapeutic outcomes.
- Advanced AI algorithms assist in accelerating stem cell research by optimizing harvesting and differentiation processes.
- AI-driven imaging and diagnostics improve the identification and monitoring of chronic diseases, such as musculoskeletal disorders, supporting targeted regenerative therapies.
- Government support includes funding and regulatory facilitation, integrating AI tools to streamline clinical trials and approvals.
- Companies like HaemaLogiX employ AI to advance immunotherapy drug development, showing promising response rates and mortality reductions.
- AI supports personalized gene therapy development and tissue engineering innovations, propelling market expansion.
Grab a sample PDF of this report: https://www.imarcgroup.com/australia-regenerative-medicine-market/requestsample
Australia Regenerative Medicine Market Growth Factors
Chronic diseases and age-related disorders drive the Australia regenerative medicine market. In 2015 around 7.3 million Australians had chronic musculoskeletal conditions that included back conditions (4.0 million), arthritis (3.7 million), and osteoporosis or osteopenia (854,000). Therefore an important demand exists within regenerative medicine to reverse how degenerative and chronic disease processes progress because clinicians need to treat these diseases that occur with prevalence and incidence.
Increased research on harvesting, differentiation of stem cells, and data on a hugely expanded range of therapeutic uses, including spinal cord injury and cardiac disease is fueling the growth of this market. As of now, patient-specific stem cell therapies have minimized the risk of immune rejection and helped in the clinical application and regulatory approval of regenerative therapies with proven results.
The market is supported by government support for research and development with the Australian government supporting biotech companies, research institutes and the potential for regenerative medicine to change how healthcare is delivered. Regulatory agencies including the Therapeutic Goods Administration (TGA) have streamlined the process and speed of approvals, creating a friendly environment for businesses and attracting foreign investment, strengthening Australia's position.
Australia Regenerative Medicine Market Segmentation
Type Insights:
- Stem Cell Therapy
- Biomaterial
- Tissue Engineering
- Others
Application Insights:
- Bone Graft Substitutes
- Osteoarticular Diseases
- Dermatology
- Cardiovascular
- Central Nervous System
- Others
End-User Insights:
- Hospitals
- Specialty Clinics
- Others
Regional Insights:
- Australia Capital Territory & New South Wales
- Victoria & Tasmania
- Queensland
- Northern Territory & Southern Australia
- Western Australia
Key Players
- HaemaLogiX
- Australian Regenerative Medicine Institute
Recent Developments & News
- May 2025: HaemaLogiX is preparing for an ASX listing aiming to raise USD 50 million to advance its immunotherapy drug KappaMab, which shows an 83% response rate and a 46% reduction in mortality risk in multiple myeloma patients, expanding its exploration into autoimmune diseases.
- March 2025: The Australian Regenerative Medicine Institute received a USD 1.7 million National Health and Medical Research Council (NHMRC) Ideas Grant for pioneering research on in utero correction of craniofacial malformations, marking substantial progress in innovative therapeutic solutions.
- July 2025: The government’s regulatory framework enhancements by the Therapeutic Goods Administration (TGA) have expedited approval processes for regenerative products, bolstering local industry growth and attracting foreign investment and partnerships.
If you require any specific information that is not covered currently within the scope of the report, we will provide the same as a part of the customization.
https://www.imarcgroup.com/request?type=report&id=33715&flag=F
About Us
IMARC Group is a global management consulting firm that helps the world’s most ambitious changemakers to create a lasting impact. The company provide a comprehensive suite of market entry and expansion services. IMARC offerings include thorough market assessment, feasibility studies, company incorporation assistance, factory setup support, regulatory approvals and licensing navigation, branding, marketing and sales strategies, competitive landscape and benchmarking analyses, pricing and cost research, and procurement research.
Contact Us
IMARC Group,
134 N 4th St. Brooklyn, NY 11249, USA,
Email: sales@imarcgroup.com,
Tel No: (D) +91 120 433 0800,
United States: +1-201971-6302
What's Your Reaction?






















































